Regulatory consulting
Crafting the regulatory strategy, establishing Quality Management System, and driving your regulated product documentation and certification efforts.
Navigate international compliance and information security to streamline your path to market
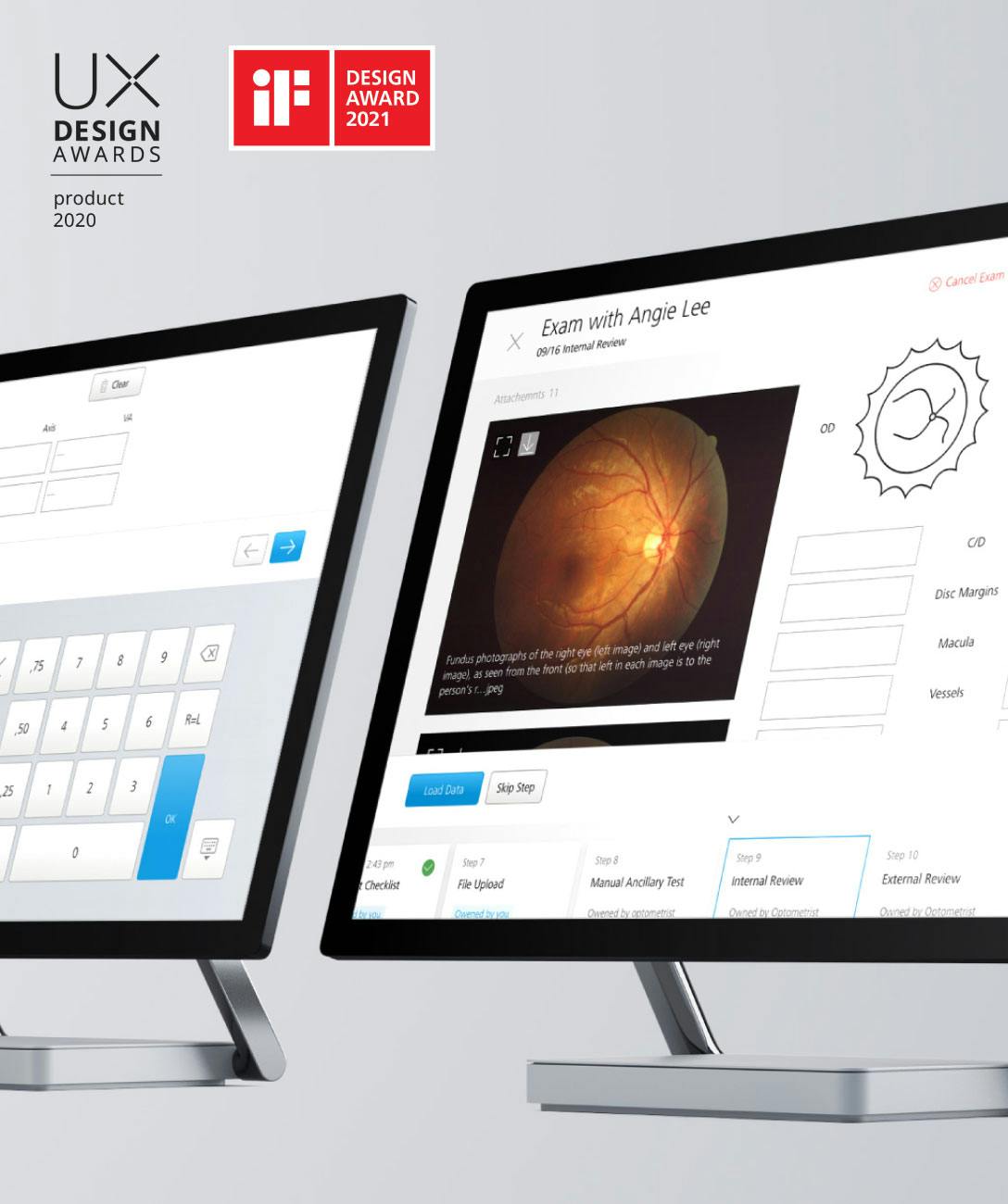
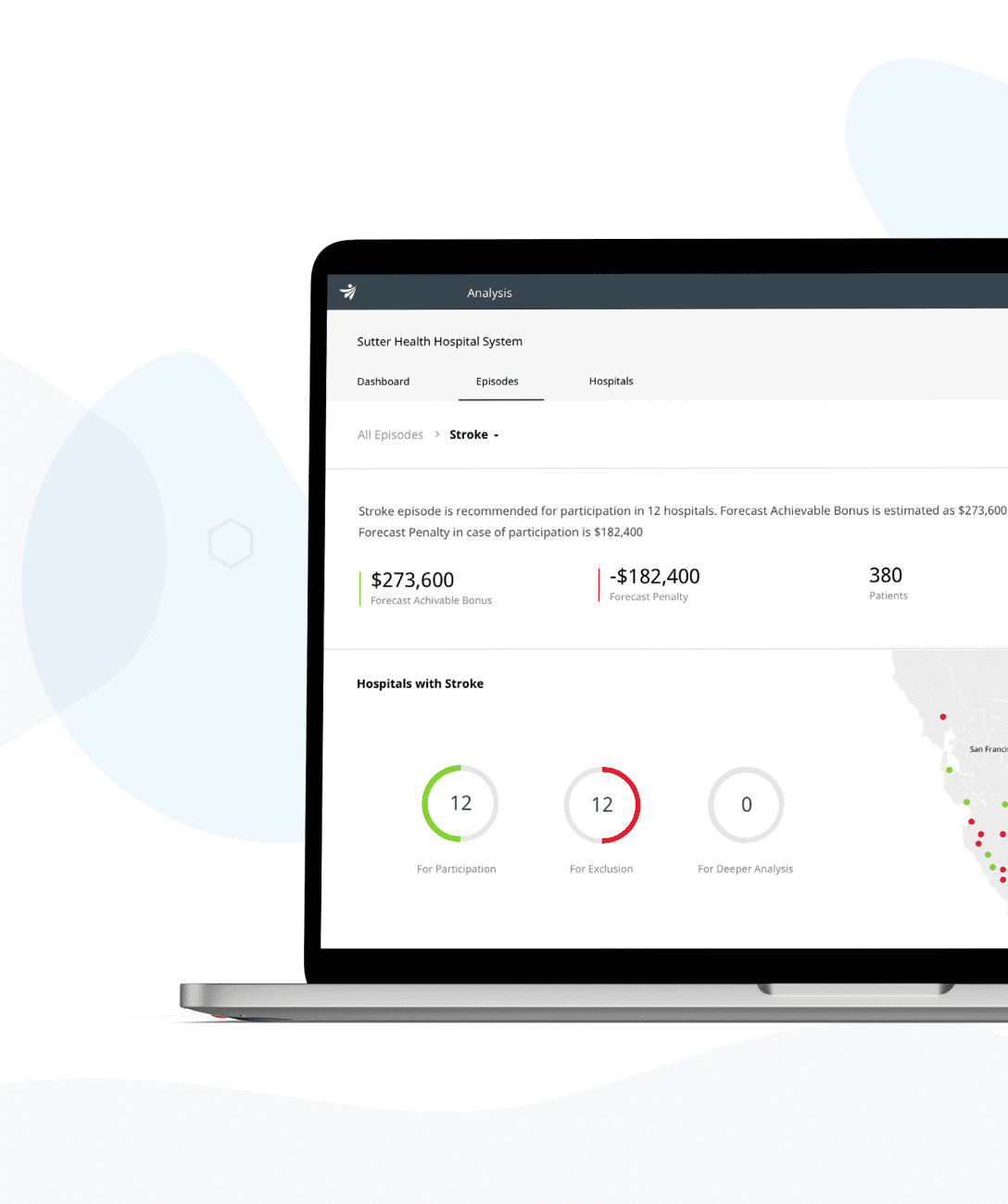
Meet regulatory and information security requirements, prepare your solutions for the entire product lifecycle, build and evolve remarkable MedTech and digital healthcare products.

We're certified and experienced healthcare technologists
What we offer
We define quality and information security management systems and guide our clients from market placement through post-market surveillance.
Regulatory strategy definition
Our unique workshop is designed to provide precise support in defining the Intended Purpose and devising an optimal regulatory strategy for your medical device.
Ready-to-use QMS knowledge base
Offering document templates as a starting point for establishing a Quality Management System in your organization, ensuring compliance with major market regulators (i.e. Swissmedic, TGA, MHRA), and based on ISO 13485, ISO 14971, and IEC 62366.
Product compliance and documentation support
Our team offers a distinctive blend of refined regulatory expertise and comprehensive software knowledge, enabling us to assist you in documenting your product and simplifying your compliance journey (in the EU, US, AU, UK, CH).
Quality manager as a service
Our regulatory experts, each boasting over a decade of experience, are ready to lead the QMS definition, implementation, training, certification and continuous improvement process.
Internal or supplier audits
Rely on our team of certified lead auditors for thorough gap assessments and QMS audits, whether for your current QMS or as a supplier control measure.
AI regulations
Elevate your team's proficiency in quality and regulatory matters with our training program. From audits to ISO 14971 risk management, ISO 13485, IEC 62304 requirements, AI regulations and more.
Ready-to-use ISMS knowledge base
Get a set of document templates to be used for establishing Information Security Management System in the organization compliant with ISO 27001:2022.
Information security officer as a service
Allow our seasoned team of regulatory experts, boasting over a decade of experience, to lead your ISMS definition, implementation, training, certification and continuous improvement processes.
Our clients say...
Contact our experts
Explore our expertise